Loss of Proteostasis: When Protein Folding Goes Wrong
Explore how the loss of proteostasis, the cell's protein quality control system, contributes to aging and age-related diseases. Learn about emerging interventions.
Table of Contents
DISCLAIMER
This article is for informational purposes only and does not constitute medical advice. The statements in this article have not been evaluated by the FDA. The information presented is based on published research and should not be used as a substitute for professional medical guidance. Consult your physician before starting any supplement or health protocol.
In the intricate machinery of life, proteins are the workhorses, performing nearly every function within a cell. From catalyzing reactions to providing structural support, transporting molecules, and transmitting signals, the proper function of these proteins is paramount for cellular health and organismal survival. However, proteins must adopt highly specific three-dimensional structures to perform their roles effectively. This complex process, known as protein folding, is meticulously managed by a sophisticated cellular quality control system called proteostasis.
Proteostasis, a portmanteau of “protein” and “homeostasis,” refers to the dynamic balance of protein synthesis, folding, trafficking, assembly, and degradation within a cell (Balch et al., 2008; PMID: 18274533). It is a fundamental cellular process that ensures the integrity and functionality of the entire proteome – the complete set of proteins expressed by an organism. As we age, however, this finely tuned system often falters, leading to a phenomenon known as the loss of proteostasis. This decline is now recognized as one of the key hallmarks of aging, contributing significantly to cellular dysfunction and the development of numerous age-related diseases.
Understanding the mechanisms behind the loss of proteostasis and exploring potential interventions to restore its balance represents a crucial frontier in longevity research. This article delves into the intricacies of the proteostasis network, examines how its efficiency diminishes with age, explores the wide-ranging consequences for health, and discusses emerging strategies to support this vital cellular process.
What is Proteostasis? A Primer on Cellular Protein Quality Control
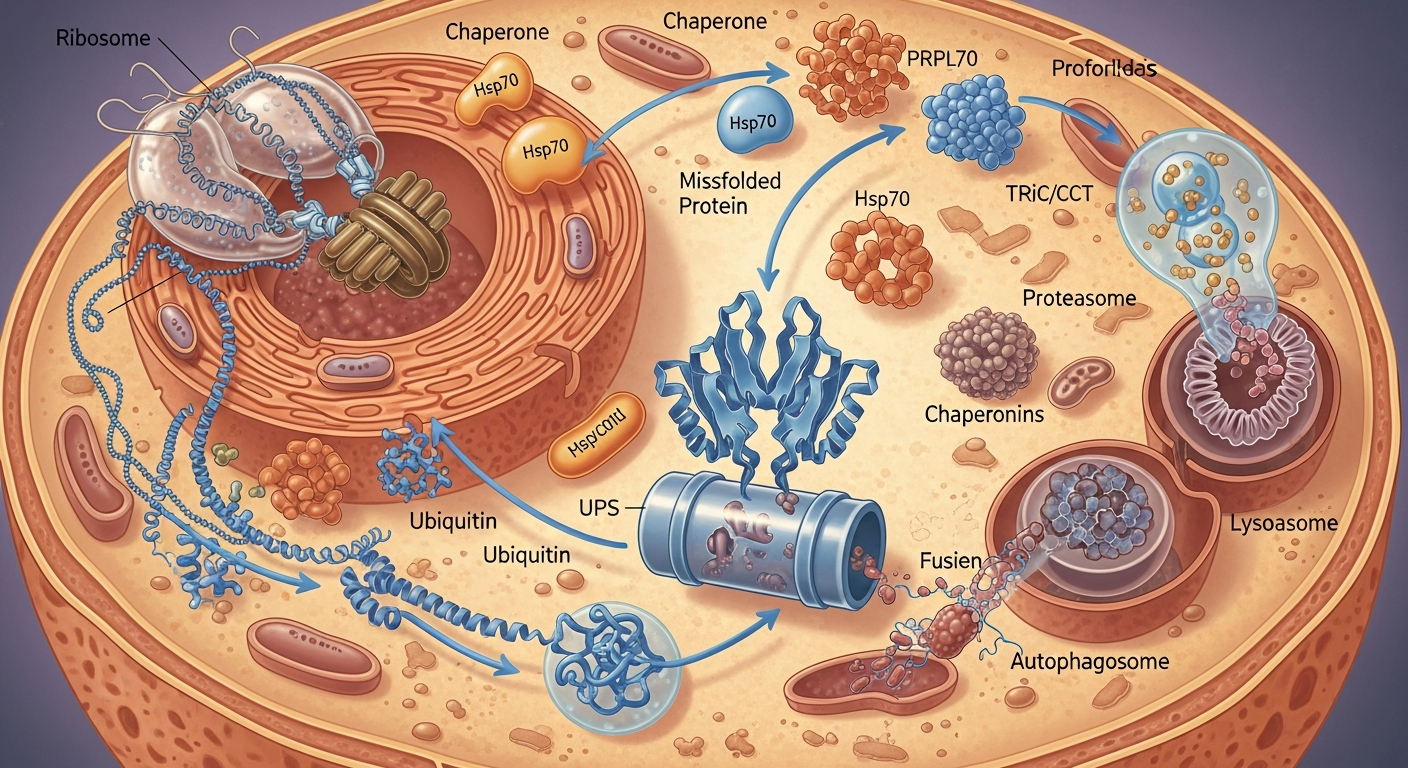
At its core, proteostasis is the cell’s overarching strategy to maintain a healthy and functional proteome. This dynamic network involves a complex interplay of molecular pathways and components that collaborate to ensure proteins are correctly folded, trafficked to their proper locations, and efficiently degraded when damaged or no longer needed.
The Proteostasis Network (PN): A Symphony of Systems
The proteostasis network (PN) can be conceptualized as an integrated system composed of three main branches:
- Protein Synthesis and Folding: Responsible for generating new proteins and assisting them in achieving their correct three-dimensional structures.
- Protein Trafficking and Assembly: Ensuring proteins reach their correct cellular compartments and, if necessary, assemble into larger functional complexes.
- Protein Degradation: Removing misfolded, damaged, or superfluous proteins to prevent their accumulation, which can be toxic to the cell.
These branches are not isolated but rather work in concert, constantly adapting to cellular stress and environmental changes.
Protein Synthesis and Folding: The Birth of a Protein
The journey of a protein begins with its synthesis on ribosomes, where messenger RNA (mRNA) is translated into a chain of amino acids. This polypeptide chain then embarks on a remarkable transformation, folding into a precise three-dimensional shape. This folding process is often spontaneous but is highly prone to errors, especially for larger and more complex proteins.
To prevent misfolding and aggregation, cells employ a specialized group of proteins known as molecular chaperones. These “helper proteins” bind to nascent polypeptide chains and partially folded proteins, guiding them towards their correct conformations and preventing them from interacting inappropriately with other proteins (Hartl et al., 2011; PMID: 22020297). Key chaperone families include:
- Heat Shock Proteins (HSPs): Such as Hsp70 and Hsp90, which are highly conserved and induced by stress, playing critical roles in protein folding, assembly, and transport.
- Chaperonins: Like the GroEL/GroES system in bacteria or TRiC/CCT in eukaryotes, which form large barrel-shaped structures where proteins can fold in an isolated environment.
Protein Degradation Pathways: Clearing the Clutter
Even with the diligent efforts of chaperones, some proteins inevitably misfold or become damaged over time. The accumulation of such aberrant proteins can lead to cellular toxicity and dysfunction. Therefore, robust degradation pathways are essential for maintaining proteostasis. The two primary degradation systems are:
-
The Ubiquitin-Proteasome System (UPS): This highly specific pathway targets individual proteins for destruction. Proteins destined for degradation are tagged with a small protein called ubiquitin, which acts as a signal for the proteasome. The proteasome is a large, multi-catalytic protein complex that recognizes ubiquitinated proteins and degrades them into smaller peptides. The UPS is crucial for regulating the levels of many short-lived regulatory proteins and for clearing misfolded proteins from the cytosol and nucleus.
-
Autophagy (Autophagosome-Lysosome Pathway): Meaning “self-eating,” autophagy is a bulk degradation process that removes larger cellular components, including damaged organelles (like mitochondria), protein aggregates, and even entire pathogens. During autophagy, a double-membraned vesicle called an autophagosome engulfs the target material. The autophagosome then fuses with a lysosome, an organelle containing powerful hydrolytic enzymes, which degrade the contents. There are several forms of autophagy, including macroautophagy (the most common), microautophagy, and chaperone-mediated autophagy (CMA), which specifically targets soluble proteins for lysosomal degradation (Cuervo & Wong, 2014; PMID: 24522851).
The precise coordination between protein synthesis, folding, and degradation ensures that the cell maintains a healthy balance of functional proteins, adapting to various physiological demands and environmental stresses.
How Does Proteostasis Decline with Age?
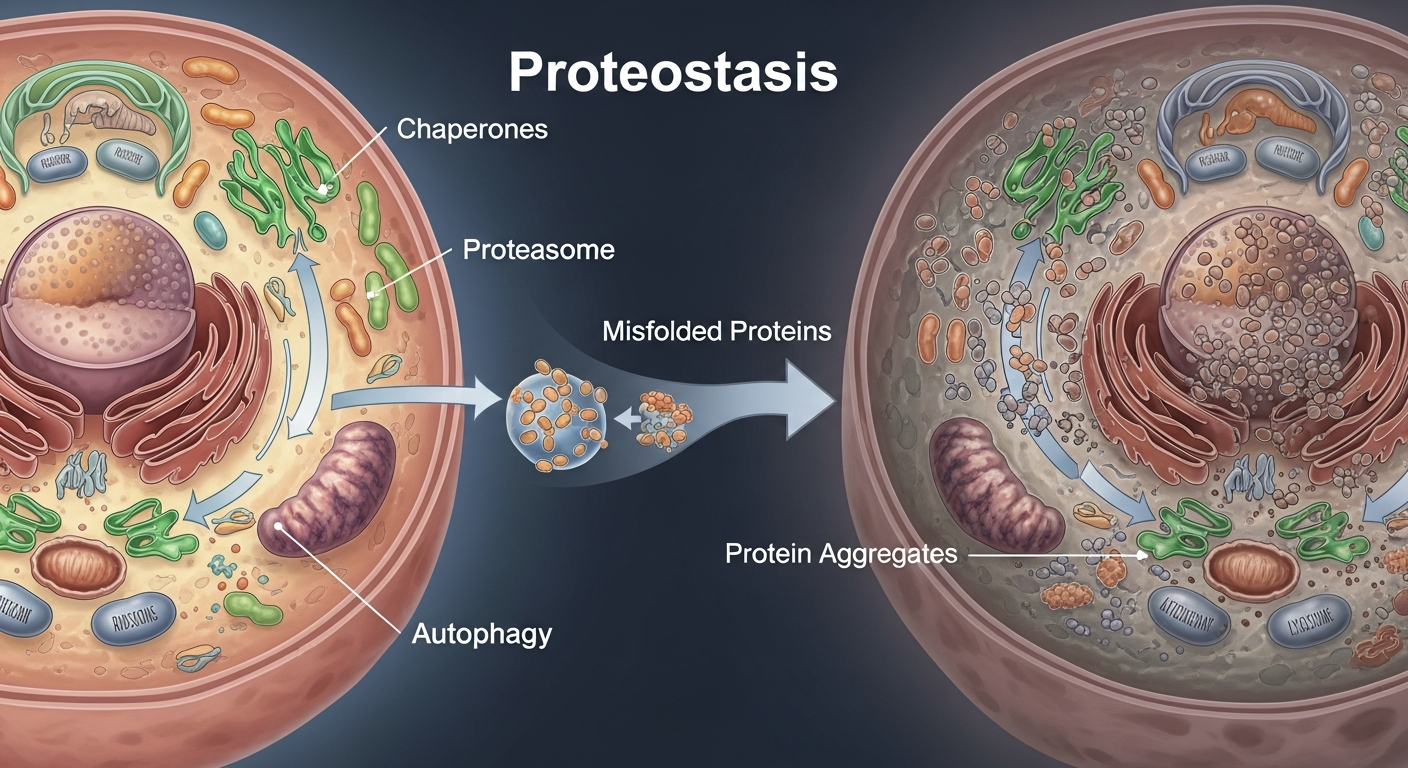
The efficiency of the proteostasis network is not static; it appears to diminish progressively with advancing age. This age-related decline is considered a fundamental contributor to the aging process itself and a common thread in many age-related pathologies (Labbadia & Morimoto, 2015; PMID: 25959140). Several factors are thought to contribute to this systemic weakening:
1. Decreased Chaperone Activity and Expression
With age, cells often exhibit a reduced capacity to synthesize and activate molecular chaperones. Studies suggest that the expression levels of key heat shock proteins, such as Hsp70 and Hsp90, may decline in various tissues of older organisms (Calderwood et al., 2010; PMID: 20689405). Furthermore, the activity of existing chaperones might be impaired due to post-translational modifications or oxidative damage. A reduced chaperone pool means fewer “helpers” are available to assist in proper protein folding, leading to an increased likelihood of misfolding and aggregation.
2. Reduced Proteasome Function
The ubiquitin-proteasome system, critical for clearing misfolded and damaged proteins, also appears to experience a decline in activity with age. Research indicates that the enzymatic activity of the proteasome may decrease in various tissues, including the brain and muscle, in older individuals and animal models (Chondrogianni et al., 2014; PMID: 24559103). This reduction can lead to a backlog of ubiquitinated proteins, resulting in their accumulation and the formation of potentially toxic aggregates.
3. Impaired Autophagy
Autophagy, particularly macroautophagy, is a vital cellular recycling process. Its efficiency is well-documented to decline with age across numerous species, from yeast to mammals (Levine & Kroemer, 2019; PMID: 31034910). This impairment can be attributed to several factors, including reduced expression of autophagy-related genes (ATGs), decreased lysosomal activity, and issues with autophagosome formation or fusion. A less efficient autophagic system means that damaged organelles and large protein aggregates, which are often too large for the proteasome, are not cleared effectively, contributing to cellular clutter and dysfunction. Chaperone-mediated autophagy (CMA) also shows a significant age-dependent decline, particularly relevant in neurodegenerative contexts (Cuervo, 2020; PMID: 32439719).
4. Increased Protein Damage and Stress
Aging is often accompanied by an increase in cellular stress, including oxidative stress (due to reactive oxygen species, ROS), inflammation, and metabolic imbalances. These stressors can directly damage proteins through oxidation, glycation, or other modifications, altering their structure and making them more prone to misfolding and aggregation. The combination of increased protein damage and a weakened proteostasis network creates a vicious cycle, accelerating the accumulation of dysfunctional proteins. For instance, advanced glycation end-products (AGEs) can directly interfere with protein structure and function, contributing to age-related tissue stiffness and impaired enzymatic activity (Semba et al., 2010; PMID: 20875245).
5. ER Stress and the Unfolded Protein Response (UPR)
The endoplasmic reticulum (ER) is where many secreted and transmembrane proteins are folded. When misfolded proteins accumulate in the ER, it triggers a stress response known as the Unfolded Protein Response (UPR). While the UPR is initially protective, aiming to restore ER proteostasis, chronic or prolonged ER stress, often seen in aging, can lead to maladaptive responses, including apoptosis (programmed cell death) (Hetz & Papa, 22018; PMID: 29358249). The ability of cells to resolve ER stress effectively appears to diminish with age, further exacerbating proteostasis imbalance.
In essence, the aging process appears to compromise the proteostasis network at multiple levels, leading to a diminished capacity to handle the constant challenge of maintaining a healthy proteome. This imbalance shifts the cellular environment towards an accumulation of misfolded and aggregated proteins, setting the stage for cellular dysfunction and disease.
The Consequences of Impaired Proteostasis in Aging and Disease
The breakdown of proteostasis is not merely a benign consequence of aging; it is increasingly recognized as a fundamental driver of cellular dysfunction and a common pathogenic mechanism underlying a wide range of age-related diseases. When the delicate balance of protein quality control is disrupted, it can lead to the accumulation of toxic protein aggregates, which interfere with normal cellular processes and can ultimately trigger cell death.
1. Neurodegenerative Diseases
Perhaps the most well-known and devastating consequences of proteostasis collapse are seen in neurodegenerative disorders. These diseases are often characterized by the accumulation of specific misfolded proteins in the brain, forming insoluble aggregates or plaques that are toxic to neurons.
- Alzheimer’s Disease: Characterized by the accumulation of amyloid-beta (Aβ) plaques and neurofibrillary tangles composed of hyperphosphorylated tau protein. Both Aβ and tau are misfolded proteins that overwhelm the proteostasis network, leading to neuronal dysfunction and loss (Soto & Castilla, 2004; PMID: 15159020).
- Parkinson’s Disease: Associated with the aggregation of alpha-synuclein into Lewy bodies within dopaminergic neurons. Impaired degradation pathways, particularly the UPS and autophagy, are thought to contribute to alpha-synuclein accumulation (Lim & Kim, 2018; PMID: 30419339).
- Huntington’s Disease: Caused by an expansion of a polyglutamine (polyQ) tract in the huntingtin protein, leading to its misfolding and aggregation. The proteostasis network struggles to clear these aggregates, resulting in neuronal damage (Rubinsztein, 2006; PMID: 16428987).
- Amyotrophic Lateral Sclerosis (ALS): Many forms of ALS involve the aggregation of proteins like TDP-43 or SOD1, often linked to deficiencies in chaperone function or degradation pathways.
In these conditions, the brain, with its highly specialized and long-lived neurons, appears particularly vulnerable to the consequences of proteostasis imbalance.
2. Metabolic Disorders
Emerging research suggests a strong link between impaired proteostasis and metabolic dysfunction, including conditions like type 2 diabetes and obesity.
- Type 2 Diabetes: The endoplasmic reticulum (ER) plays a critical role in insulin production in pancreatic beta-cells. Chronic ER stress, a form of proteostasis imbalance, can impair insulin secretion and contribute to beta-cell dysfunction and death, a hallmark of type 2 diabetes (Ozcan & Tabas, 2010; PMID: 20463773). Misfolding of proteins involved in glucose metabolism or insulin signaling can also contribute.
- Obesity: Adipose tissue proteostasis appears to be crucial for its healthy expansion and function. Dysregulation may contribute to inflammation and insulin resistance associated with obesity.
3. Cardiovascular Disease
The heart and vasculature are also susceptible to proteostasis decline.
- Cardiac Amyloidosis: A condition where misfolded proteins (often transthyretin) accumulate in the heart muscle, leading to stiffness and impaired pumping function.
- Atherosclerosis: The accumulation of misfolded proteins and impaired cellular waste removal in vascular cells may contribute to the inflammatory processes and plaque formation characteristic of atherosclerosis.
4. Cancer
While often associated with cell proliferation, proteostasis also plays a complex role in cancer. Cancer cells often exhibit an altered proteostasis network, sometimes relying on specific chaperones to survive stress or resist chemotherapy. However, severe proteostasis collapse can also induce cancer cell death, making it a potential therapeutic target (Santagata & Lindquist, 22011; PMID: 21915979).
The pervasive nature of proteostasis decline across these diverse disease states underscores its fundamental importance to healthy aging. Maintaining a robust proteostasis network appears to be a critical factor in extending healthspan and preventing age-related pathologies.
Table: Key Components of the Proteostasis Network and Age-Related Changes
This table summarizes the main elements of the proteostasis network and how their function may be affected during aging.
| Proteostasis Component | Primary Function | Age-Related Change (Research Suggests) | Potential Consequences in Aging
Sources
- The proteostasis network in aging and disease(2011)
- Chaperone-mediated autophagy in aging and neurodegeneration(2020)
- Autophagy and aging: a complex interplay(2021)
- Exercise and the regulation of proteostasis(2018)
- Nutritional interventions to enhance proteostasis and healthy aging(2022)
- Pharmacological targeting of proteostasis for healthy aging(2021)
- The unfolded protein response in aging and age-related diseases(2018)
Stay Updated on Longevity Science
Weekly research digests. No spam, unsubscribe anytime.
Related Articles

Autophagy and Aging: Your Body's Cellular Cleanup System
Explore autophagy, your body's cellular cleanup system, and its critical role in aging and longevity. Research suggests modulating autophagy may support cellular health.
12 min read
Epigenetic Alterations: How Your Genes Age Without Changing
Understand how epigenetic alterations drive aging through DNA methylation changes, histone modifications, and chromatin remodeling -- and what research suggests.
14 min read
Genomic Instability and Aging: How DNA Damage Drives the Aging Process
Explore how genomic instability contributes to aging, the role of DNA damage and repair mechanisms, and emerging research on protecting genomic integrity.
15 min read